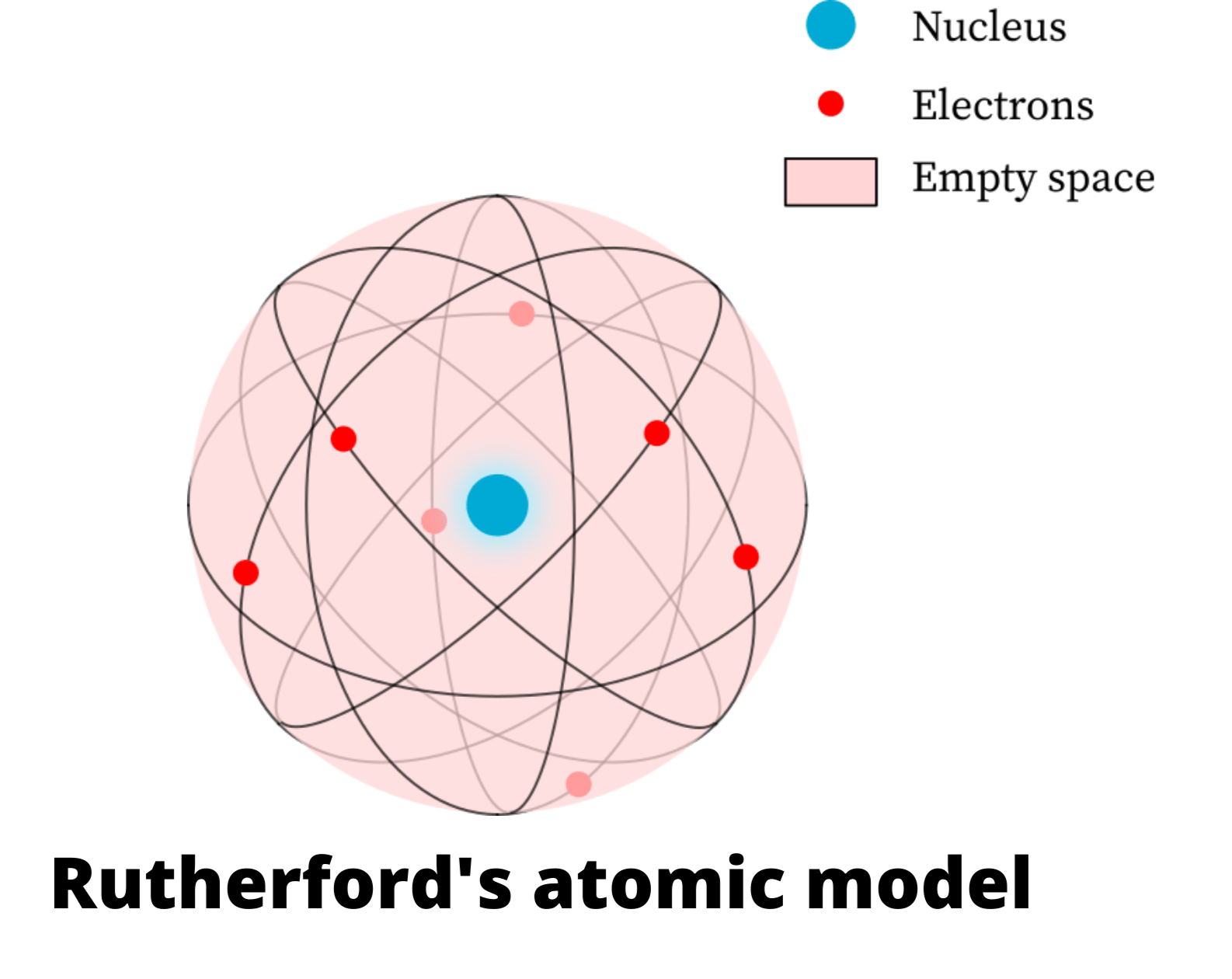
He established that, for this to occur, an atom should have the same quantity of negative and positive charges. Thomson knew that the atoms were electrically neutral. Crookes named the emission ' cathode rays'.Īfter the cathode ray experiments, Sir Joseph John Thomson established that the emitted ray was formed by negative charges, because they were attracted by the positive pole. When creating a vacuum in the tube, a light discharge can be seen that goes from the cathode (negatively-charged electrode) to the anode (positively-charged electrode). In 1850, Sir William Crookes constructed a ' discharge tube', that is a glass tube with the air removed and metallic electrodes at its ends, connected to a high- voltage source. Thomson's atomic model Schematic representation of the Thomson model. Together, these were good evidence that atoms existed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
