 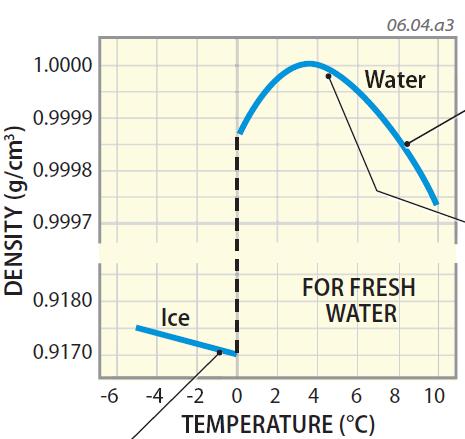
Therefore, the correct answer is option-2: the density of water increases when heated from 0☌ to 4☌. 
As a result, the molecules become less organized and spaced out, leading to a decrease in density. When water is heated beyond this temperature, the additional thermal energy disrupts the hydrogen bonds. However, at 4☌, the hydrogen bonds between water molecules become more organized, and this causes a dense arrangement of molecules, resulting in the maximum density. Solve any question of Thermal Properties Of Matter with:. Water has the maximum density of 1g/cm3 only when it is pure water. Specific gravity is defined as the ratio of the density of a fluid to the density of water at a reference temperature. When the temperature changes from either greater or less than 4 degrees, the density will become less than 1g/cm3. The rationale for finding the density and specific weight of water at 4C in the foregoing discussion is that 4C is a reference temperature on which specific gravity is based. At temperatures below 4☌, the increased molecular motion causes the water molecules to spread out, leading to a decrease in density. Suppose I have 1kg of water at 4°rm C.So its density is 1rm: g/cm³. Water has its maximum density of 1g/cm3 or 1000kg/m3 at 4C. Beyond this point, when water is heated further, its density starts to decrease again.Īs water is heated, the molecules gain thermal energy and begin to move more rapidly. This is because water is unique in the sense that its density decreases as it gets colder, until it reaches its maximum density at approximately 4☌. Thus, the density of water is a maximum at 4 ☌.When water is heated from 0☌ to 4☌, its density increases. Cluster formation is the bigger effect, so the density starts to decrease. The molecules are still slowing down and coming closer together, but the formation of clusters makes the molecules be further apart. As the temperature of warm water decreases, the water molecules slow down and the density increases. The molecules of the water are closer together, and this increases the density of the liquid. The literature reports values of 1.028 g/cm 3 for the density of microcrystalline cellulose, and. The clusters still have empty spaces, so they decrease the density of the liquid. The results of the hydrogel density are based on a water density of 0.997 g/cm 3 at 4 C 47. These clusters are free to move relative to each other, so water is still liquid. At temperatures near 0 ☌, the water still contains many ice-like clusters. S tate whether the object will float or sink in water. An object of mass 100 g has a volume of 20 c m 3. The latent heat of vapourisation of water is 2.25 10 6 J/kg. The density decreases as temperature increases. Water has its maximum density of 1g/cm 3 or 1000 kg/m 3 at 4 degrees Celsius. The increase in internal energy of 1 kg of water at 100 0 C when it is converted into steam at the same temperture and at 1 atm (100 kPa) will be: (The density of water and steam are 1000 k g / m 3 and 0.6 k g / m 3 respectively. The density of water increases with decreasing temperature, reaching a maximum at 4.0 C, 4.0 C, and then decreases as the temperature falls below 4.0 C 4.0 C. Table 14.2 shows the density of water in various phases and temperature. At temperatures well above freezing, the molecules move faster and get further apart. The density of solids and liquids normally increase with decreasing temperature. When ice melts to liquid water density of the liquid increases as the structure collapses. 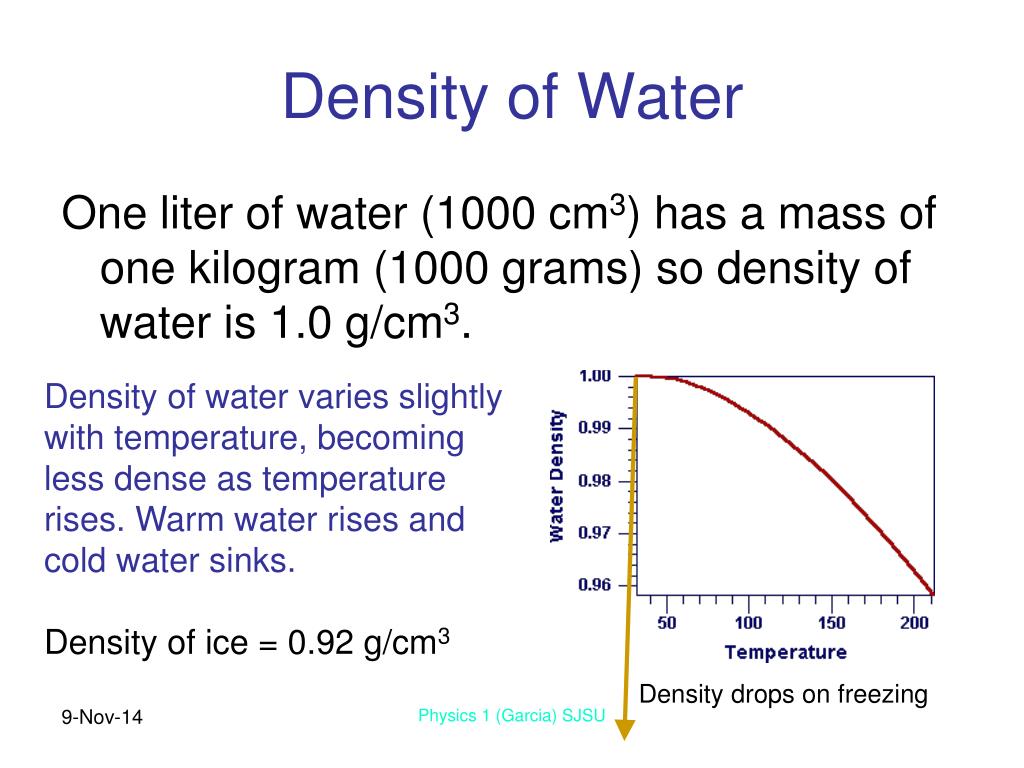
The maximum density of water at 4☌ is 1 g/cm³ or 1000 kg/m³. Cluster formation is the bigger effect, so the density starts to decrease. We know that the density of water is maximum at 4☌. What is water's density at 94 C Assume a constant coefficient of volume expansion. As explained above due to anomalous behaviour of water when water is heated from 0° C to 10° C, then its volume first decreases and then increases although mass remains constant. This means that water has a maximum density (1 g/cm3) at 4☌. The water molecules in ice are present in a crystal lattice that has lot of empty space. As the temperature of warm water decreases, the water molecules slow down and the density increases. The density of water at 4C is 1.00×103kg/m3. Below 4 ☌, the volume increases, and therefore the density decreases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
