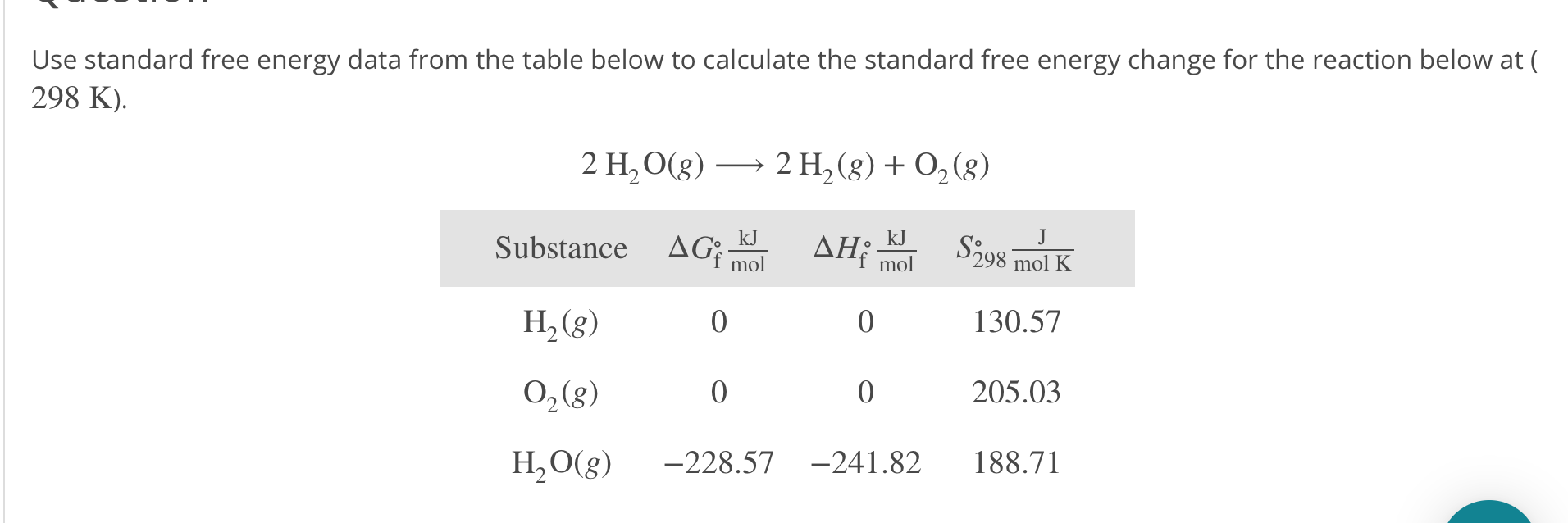
EXAMPLE 2 Determine the standard entropy change for the. By expanding consideration of entropy changes to include the surroundings, we may reach a significant conclusion regarding the relation between this property and spontaneity. a result of the Third Law of Thermodynamics, which defines a zero for entropy and thus allows us to calculate absolute entropy values. Processes that involve an increase in entropy of the system (Δ S > 0) are very often spontaneous however, examples to the contrary are plentiful. Calculation of absolute molecular entropies and heat capacities made simple. 5. 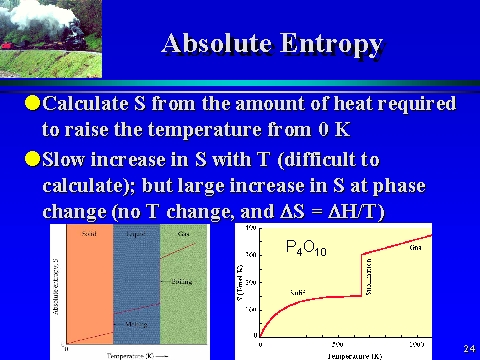
In the quest to identify a property that may reliably predict the spontaneity of a process, we have identified a very promising candidate: entropy. But this leaves us in a rather uncomfortable position. Now that we have calculated the absolute entropy at a given temperature and pressure, we can calculate the increase in the Helmholtz and Gibbs functions from equations 12.9.9 and 12.9.11.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
